Quick answer
Zepbound (tirzepatide) produces more average weight loss than Wegovy (semaglutide) in clinical trials, including the first direct head-to-head study, SURMOUNT-5, published in 2025. Tirzepatide is a dual GIP/GLP-1 agonist; semaglutide is a single GLP-1 agonist. Both are once-weekly injections, both require titration over months, and both carry similar gastrointestinal side effects. Zepbound has a lower list price ($1,086 vs $1,349) and a slightly cheaper self-pay floor ($349 via LillyDirect vials vs $499 via NovoCare). Insurance coverage is comparable. The "better" drug depends on your tolerance, your insurance, and what your prescriber recommends — not on the trial averages alone.
The short version is that Zepbound usually wins on the scale, but Wegovy has years more real-world data, a longer safety record, an established cardiovascular indication, and a self-pay price that is identical for every dose. For some patients, that combination matters more than the few extra percentage points of weight loss.
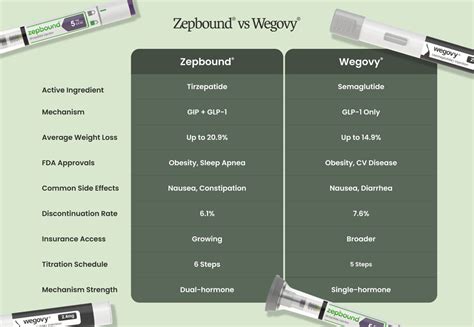
Side-by-side comparison
Here are the FDA-label facts on both drugs in one table. We pull this data from each manufacturer's prescribing information and the original clinical trial reports.
| Wegovy | Zepbound | |
|---|---|---|
| Brand name | Wegovy | Zepbound |
| Active ingredient | semaglutide | tirzepatide |
| Drug class | GLP-1 receptor agonist | GIP/GLP-1 dual agonist |
| FDA indication | Chronic weight management | Chronic weight management |
| How it’s taken | Subcutaneous injection, once weekly | Subcutaneous injection, once weekly |
| Maximum dose | 2.4 mg weekly | 15 mg weekly |
| Average weight loss | 14.9% body weight at 68 weeks (STEP 1 trial) | 20.9% body weight at 72 weeks (SURMOUNT-1 trial, 15 mg dose) |
| FDA approval | 2021 | 2023 |
| List price | $1349/mo retail, $499/mo NovoCare cash pay | $1086/mo retail, $349-$499/mo LillyDirect self-pay |
A few things stand out. Both are weekly subcutaneous injections. Both are FDA-approved for chronic weight management with similar BMI criteria. Wegovy was approved in 2021; Zepbound followed in November 2023, making it the newer of the two. The maximum dose looks dramatically different — 2.4 mg for Wegovy versus 15 mg for Zepbound — but the milligram numbers are not comparable across drugs because the molecules and potencies are different.
Different active ingredients: semaglutide vs tirzepatide
The most important difference between the two drugs is what is actually inside the pen.
- Wegovy contains semaglutide, a synthetic peptide that closely mimics natural human GLP-1 (glucagon-like peptide-1). It is engineered with a fatty acid side chain that lets it bind to albumin in the blood, extending its half-life from minutes to about a week. Novo Nordisk has been developing semaglutide molecules since the early 2010s, and it is also the active ingredient in Ozempic and Rybelsus.
- Zepbound contains tirzepatide, a synthetic peptide engineered by Eli Lilly that is structurally distinct from semaglutide. Unlike semaglutide, tirzepatide is a single molecule that binds to two different incretin hormone receptors at once. It is the same active ingredient as Mounjaro, Lilly's diabetes brand.
This is why a pharmacist cannot substitute Zepbound for Wegovy or vice versa, even though both treat the same condition. They are different molecules made by different manufacturers with different patents, different clinical trial programs, and different supply chains.
For a deeper look at semaglutide on its own, see our main Wegovy guide. For more on the broader landscape, see all GLP-1 weight loss drugs compared.
Single vs dual agonist: what that actually means
You will see semaglutide described as a "single agonist" and tirzepatide as a "dual agonist". Here is what those labels actually describe at the cellular level.
When you eat, your gut releases two hormones almost immediately: GLP-1 (glucagon-like peptide-1) and GIP (glucose-dependent insulinotropic polypeptide). Both are called incretin hormones because they help signal that food has arrived. GLP-1 slows stomach emptying, suppresses glucagon, stimulates insulin, and reduces hunger via the brain. GIP also stimulates insulin release, plays a role in fat tissue metabolism, and may modulate nausea pathways.
- Semaglutide (Wegovy) binds only to the GLP-1 receptor. It activates one of the two incretin pathways. This is enough to drive substantial weight loss — STEP 1 saw 14.9% average loss — and it has been the gold standard since 2021.
- Tirzepatide (Zepbound) is one molecule that binds to both the GIP receptor and the GLP-1 receptor. By activating both incretin pathways simultaneously, it appears to produce a stronger combined signal — more appetite suppression, more weight loss, and possibly some offsetting effect on nausea from the GIP component.
Researchers have hypothesized for years that hitting more than one incretin receptor would produce better outcomes than GLP-1 alone. Tirzepatide was the first molecule to test that hypothesis at scale, and the trial data supports it: more weight loss, better A1C reduction in diabetes trials, and a similar safety profile. Whether triple agonists (such as the experimental drug retatrutide) will go even further is the next big question.

Weight loss effectiveness: clinical trial data
The two drugs have different lead trials. Here is what the original studies showed, before any direct comparison.
STEP 1 — Wegovy
STEP 1 was a 68-week, double-blind, randomized trial of semaglutide 2.4 mg weekly in 1,961 adults with obesity or overweight without diabetes. Participants on semaglutide lost an average of 14.9% of their body weight, compared to 2.4% in the placebo group. About 86% of patients on semaglutide lost at least 5% of body weight, and roughly one in three lost more than 20%. Results were published in the New England Journal of Medicine in 2021.
SURMOUNT-1 — Zepbound
SURMOUNT-1 was a 72-week, double-blind, randomized trial of tirzepatide in 2,539 adults with obesity or overweight without diabetes. Participants were randomized to one of three tirzepatide doses (5, 10, or 15 mg weekly) or placebo. At the highest 15 mg dose, average weight loss was 20.9%; at 10 mg it was 19.5%; at 5 mg it was 15.0%. Roughly 91% of patients lost at least 5% of body weight, and more than half on the 15 mg dose lost 20% or more. Results were published in the New England Journal of Medicine in 2022.
| Trial | Drug & dose | Duration | Avg weight loss | ≥5% loss |
|---|---|---|---|---|
| STEP 1 | Semaglutide 2.4 mg (Wegovy) | 68 weeks | 14.9% | ~86% |
| SURMOUNT-1 | Tirzepatide 5 mg (Zepbound) | 72 weeks | 15.0% | ~85% |
| SURMOUNT-1 | Tirzepatide 10 mg | 72 weeks | 19.5% | ~89% |
| SURMOUNT-1 | Tirzepatide 15 mg | 72 weeks | 20.9% | ~91% |
| STEP 1 / SURMOUNT-1 | Placebo | ~70 weeks | 2.4-3.1% | ~30% |
Importantly, the lowest dose of tirzepatide (5 mg) produced about the same average weight loss as the maximum dose of semaglutide (2.4 mg). The added benefit of Zepbound shows up at the higher doses, where semaglutide has no equivalent.
SURMOUNT-5 head-to-head results (2025)
For years, the comparison between Wegovy and Zepbound was indirect — researchers had to compare across two different trials with two different patient populations. That changed in 2025 with the publication of SURMOUNT-5, the first direct, randomized, head-to-head trial of tirzepatide versus semaglutide in adults with obesity.
SURMOUNT-5 was a 72-week, open-label, randomized trial of 751 adults with obesity (without type 2 diabetes), randomized to either tirzepatide (titrated to a maximum tolerated dose of 10 or 15 mg) or semaglutide (titrated to a maximum tolerated dose of 1.7 or 2.4 mg). It was sponsored by Eli Lilly. Results were published in the New England Journal of Medicine in May 2025.
| Outcome at 72 weeks | Tirzepatide (Zepbound) | Semaglutide (Wegovy) | Difference |
|---|---|---|---|
| Average body weight change | -20.2% | -13.7% | -6.5 percentage points (favoring tirzepatide) |
| Patients losing ≥10% | ~82% | ~60% | +22 pp |
| Patients losing ≥15% | ~65% | ~40% | +25 pp |
| Patients losing ≥25% | ~32% | ~16% | +16 pp |
| Waist circumference reduction | -18.4 cm | -13.0 cm | -5.4 cm (favoring tirzepatide) |
Tirzepatide beat semaglutide on every prespecified weight outcome. The average difference of about 6.5 percentage points of body weight is meaningful clinically — for a 220 lb starting weight, it works out to roughly 14 extra pounds lost on tirzepatide over the same time period.
The trial had limitations. It was open-label (patients knew which drug they were taking) and Lilly-funded, which means it should be read with the usual scrutiny applied to industry-sponsored head-to-head trials. The semaglutide group lost slightly less than the original STEP 1 average (13.7% vs 14.9%), which may reflect the open-label design or the different patient mix. Still, the direction and magnitude of the result were consistent with everything researchers expected, and the study moved the needle on professional guidelines.
Side effects compared
The two drugs have very similar safety profiles. Most side effects are gastrointestinal and most show up during the first few months of dose escalation, then ease as the body adapts. Severe or treatment-discontinuing side effects are uncommon for both.
| Side effect (any grade) | Wegovy 2.4 mg (STEP 1) | Zepbound 15 mg (SURMOUNT-1) |
|---|---|---|
| Nausea | ~44% | ~29% |
| Diarrhea | ~32% | ~23% |
| Vomiting | ~25% | ~13% |
| Constipation | ~24% | ~17% |
| Abdominal pain | ~20% | ~10% |
| Fatigue | ~11% | ~7% |
| Discontinued due to AE | ~7% | ~6% |
On the surface, semaglutide reported higher rates of GI side effects than tirzepatide in their respective trials. But these numbers are not from a head-to-head comparison and the trial designs differed, so they should be read as approximate, not definitive. SURMOUNT-5 reported more comparable GI rates between the two drugs, with tirzepatide producing slightly more nausea at the highest doses. The honest summary is that both drugs cause similar GI symptoms, both are easier to tolerate when titration is slow, and the "easier on my stomach" answer varies by individual.
Both drugs share the same boxed warning for thyroid C-cell tumors observed in rodent studies. Both are contraindicated in patients with a personal or family history of medullary thyroid carcinoma or multiple endocrine neoplasia syndrome type 2 (MEN 2). For a deeper look at the full side effect list and what to do about each, see our Wegovy side effects guide.
Severe abdominal pain that radiates to the back (possible pancreatitis), severe vomiting and dehydration, yellowing of the skin or eyes (possible gallbladder issue), or a new neck lump or persistent hoarseness. These warnings apply equally to Wegovy and Zepbound.
Dosing and titration
Both drugs are once-weekly subcutaneous injections, and both follow a slow upward dose titration to limit nausea. The dose strengths look very different on paper, but each manufacturer's titration ladder is designed for the same outcome: get to a maintenance dose over roughly four to five months.
| Week range | Wegovy (semaglutide) | Zepbound (tirzepatide) |
|---|---|---|
| Weeks 1-4 | 0.25 mg | 2.5 mg |
| Weeks 5-8 | 0.5 mg | 5 mg |
| Weeks 9-12 | 1.0 mg | 7.5 mg |
| Weeks 13-16 | 1.7 mg | 10 mg |
| Week 17+ | 2.4 mg (maintenance) | 12.5 mg or 15 mg (maintenance) |
A few practical differences. Wegovy comes only in pre-filled, pre-measured pens — no vials, no measuring. Zepbound is available in both pens and, since 2024, in single-use vials sold through LillyDirect at a lower self-pay price. The vials require drawing up the dose with a syringe, which is slightly more complex but cuts the price meaningfully for cash-pay patients. For a step-by-step Wegovy titration walkthrough, see our Wegovy dosing guide.
Many providers will hold a dose for an extra month if side effects are bothersome, regardless of which drug. Some patients reach their weight goal at 1.7 mg of Wegovy or 7.5 mg of Zepbound and never need to titrate to the maximum.
Cost comparison: LillyDirect vs NovoCare
On sticker price, Zepbound is meaningfully cheaper than Wegovy. On self-pay programs, the gap is narrower but still real.
| Pricing channel | Wegovy (semaglutide) | Zepbound (tirzepatide) |
|---|---|---|
| Retail list price | ~$1,349/mo | ~$1,086/mo |
| Manufacturer self-pay | $499/mo (NovoCare, all doses) | $349-$499/mo (LillyDirect vials) |
| With commercial insurance + savings card | $0-$25/mo (NovoCare Savings Card) | $0-$25/mo (Zepbound Savings Card) |
| Medicare (weight loss) | Not covered | Not covered |
| Medicare (other indication) | May be covered for CV indication (2024+) | May be covered for sleep apnea indication (2024+) |
| Pen vs vial | Pen only | Pen or vial |
The big change in 2024-2025 was that both manufacturers responded to political and competitive pressure by launching direct-to-consumer self-pay programs. Eli Lilly went first with LillyDirect, offering Zepbound vials at $349 for the lowest dose and $499 for higher doses. Novo Nordisk followed with NovoCare at a flat $499 for all Wegovy doses. For uninsured patients, this collapsed roughly two-thirds off the retail price.
For commercially insured patients whose plan covers either drug, both manufacturer savings cards can drop monthly copays to $0-$25. Coverage criteria are similar: BMI ≥ 30 or ≥ 27 with a comorbidity, prior authorization, sometimes step therapy. If you want a detailed cost breakdown, see our Wegovy cost guide.
The lowest LillyDirect price applies to the 2.5 mg starting vial. Higher doses are priced at $499. By contrast, NovoCare charges $499 for any Wegovy dose, including the starting 0.25 mg dose. So NovoCare is more expensive at the start but the same price at maintenance.
Insurance coverage differences
Insurance coverage for Wegovy and Zepbound is structurally similar. Both are now covered by many commercial plans, both are typically placed on Tier 3 (non-preferred brand), both usually require prior authorization, and both have manufacturer savings cards that can drop copays to almost nothing for eligible commercial patients. The differences come down to which specific drug is on your plan's preferred formulary that year.
- Commercial / employer plans: Most large employer plans now cover at least one of the two, and a growing number cover both. Some plans place Wegovy as the preferred GLP-1 and require step therapy before Zepbound; others do the opposite. Check your formulary or call your benefits line.
- Medicare Part D: By statute, Medicare cannot cover drugs prescribed solely for weight loss. However, both drugs now have non-weight-loss FDA indications that Part D can cover: Wegovy for cardiovascular event reduction (approved March 2024) and Zepbound for moderate-to-severe obstructive sleep apnea in adults with obesity (approved December 2024). If you have one of those conditions, ask your prescriber about coverage under the cardiovascular or sleep apnea indication.
- Medicaid: Coverage varies by state. As of early 2026, roughly 13 states cover GLP-1 weight loss drugs through Medicaid. Most states cover neither. Check your state's preferred drug list for both Wegovy and Zepbound.
- Tricare and VA: Generally do not cover either drug for weight loss. Limited coverage may exist for specific clinical scenarios.
A 2025 industry survey found that PBMs were beginning to "play favorites" between the two drugs, sometimes negotiating exclusive contracts that favored one. If your insurance suddenly stops covering one, the other may now be the preferred option on the same plan. That is a real, practical reason to know about both before you start.
Which should you choose? A decision framework
There is no universally correct answer — but there are some clear cases where one drug fits better than the other. Think of these as tiebreakers, not rules.
Choose Wegovy if...
- You have established cardiovascular disease and want a drug that is FDA-approved to reduce cardiovascular events.
- Your insurance covers Wegovy but not Zepbound.
- You prefer a single, fixed self-pay price for any dose (NovoCare's $499 is the same all the way up).
- You only want pen-based dosing and do not want to deal with vials and syringes.
- You have already tried Wegovy and tolerated it well — there may be no reason to switch.
- You value the longer real-world track record. Wegovy has been in use for weight management since 2021; Zepbound since late 2023.
Choose Zepbound if...
- You want maximum average weight loss based on the trial data, including the SURMOUNT-5 head-to-head.
- You have moderate-to-severe obstructive sleep apnea and obesity, and want a drug FDA-approved for that condition.
- Your insurance covers Zepbound but not Wegovy, or it is on a lower tier on your plan.
- You are paying cash and want the lowest entry price ($349/month for LillyDirect vials at the starting dose).
- You have not responded well to Wegovy and your prescriber recommends a switch.
- You want to try the dual-agonist mechanism — newer biology, more incretin pathways activated.
Either drug is fine if...
- You qualify clinically (BMI ≥ 30, or BMI ≥ 27 with a comorbidity), have insurance coverage for both, and have no preference on mechanism. In that case, your prescriber's experience and your local supply situation are probably the biggest factors.
- Your weight loss goal is moderate (under 15% of body weight). Both drugs comfortably cover that range.
- You want to use a manufacturer self-pay program to skip insurance entirely. Both offer one.
The right answer for most people involves a real conversation with a prescriber. Trial averages do not predict individual results, and the difference between "this drug works for me" and "this drug doesn't" is often something the data cannot tell you up front.
Can you switch between Wegovy and Zepbound?
Yes. Switching from one to the other is common and is generally well tolerated when done correctly.
There is no formal washout requirement in either prescribing label, but most providers wait at least one week after the last dose of the old drug before starting the new one — that is, you simply skip your next scheduled injection and start the new drug on what would have been your next dose day. This avoids any overlap.
The more important rule is to restart titration at the lowest dose of the new drug, even if you were already at maintenance on the old one. Going straight from 2.4 mg of Wegovy to 15 mg of Zepbound is a recipe for severe nausea, even though it might seem logical based on "I was already at the top dose". The receptors and pharmacokinetics are different enough that your tolerance to one does not transfer to the other. Most providers start switchers at 2.5 mg of Zepbound (or 0.25 mg of Wegovy if going the other way) and titrate normally over 16-20 weeks.
Reasons people switch include: lack of weight loss response on the first drug, intolerable side effects, supply shortages, insurance formulary changes, or wanting to try the dual-agonist mechanism after plateauing on a single agonist. There is no medical penalty for switching — it is one of the more flexible decisions in chronic obesity management.
The bottom line
Wegovy and Zepbound are the two most effective FDA-approved weight loss medications available in 2026. They share a class (incretin-based therapies), a delivery method (weekly subcutaneous injection), and a similar safety profile. They differ on the molecule (single GLP-1 vs dual GIP/GLP-1), the average weight loss in trials (about 6.5 percentage points in favor of Zepbound based on SURMOUNT-5), and on a handful of pricing and indication details.
For most people, the right choice depends less on which drug "wins" on average and more on which one your insurance covers, which one your body tolerates, and which one your prescriber feels confident managing. Both are excellent tools when used as directed, and switching between them is straightforward if the first choice does not fit.
To go deeper:
- Wegovy: complete patient guide — the full pillar overview of semaglutide
- Wegovy cost in 2026 — pricing, savings cards, and self-pay programs
- Wegovy side effects — frequencies, timelines, and management
- Wegovy dosing schedule — week-by-week titration walkthrough
- Wegovy vs Ozempic — same molecule, different indication
- All GLP-1 weight loss drugs compared — Wegovy, Zepbound, Saxenda, and more
Frequently Asked Questions
Is Zepbound better than Wegovy?
In head-to-head trial data, Zepbound (tirzepatide) produces more weight loss on average than Wegovy (semaglutide). In the SURMOUNT-5 trial published in 2025 — the first direct comparison of the two — patients on tirzepatide lost about 20.2% of their body weight at 72 weeks, compared to about 13.7% on semaglutide. That said, "better" depends on side effects, cost, insurance coverage, and individual response. Many patients do extremely well on Wegovy and never need to consider switching.
Is Wegovy a tirzepatide?
No. Wegovy is semaglutide, a single-receptor GLP-1 agonist made by Novo Nordisk. Tirzepatide is the active ingredient in Zepbound and Mounjaro, both made by Eli Lilly. Tirzepatide activates two receptors (GIP and GLP-1) instead of one, which is why it is sometimes called a "dual agonist" or "twincretin". The two drugs are in related but distinct classes.
What is the main difference between Wegovy and Zepbound?
The main difference is the active ingredient and how it works. Wegovy contains semaglutide, which activates only the GLP-1 receptor. Zepbound contains tirzepatide, which activates both the GLP-1 receptor and the GIP (glucose-dependent insulinotropic polypeptide) receptor. Activating two receptors at once appears to produce greater appetite suppression and more weight loss in clinical trials, though it does not work that way for every patient.
How does Zepbound vs Wegovy cost compare?
Zepbound has a lower retail list price than Wegovy: about $1,086 per month for Zepbound versus $1,349 per month for Wegovy. Self-pay programs make both more affordable: LillyDirect offers Zepbound vials at $349 to $499 per month, while NovoCare offers Wegovy at $499 per month for any dose. With commercial insurance and a manufacturer savings card, copays for both drugs typically fall to $0 to $25 per month for eligible patients.
Are the side effects of Zepbound vs Wegovy the same?
The side effect profiles are very similar. Both drugs commonly cause nausea, diarrhea, vomiting, constipation, abdominal pain, and fatigue, especially during dose escalation. Tirzepatide may produce slightly higher rates of GI side effects at top doses in some trials, but the difference is small and most patients on either drug see symptoms ease after the first few weeks. Both carry a boxed warning for thyroid C-cell tumors observed in rodent studies.
Can you switch from Wegovy to Zepbound?
Yes, switching is common and generally straightforward, though it should be done with a prescriber. There is no formal washout period required, but most providers recommend waiting one week from your last Wegovy dose before starting Zepbound. You typically restart titration at the lowest Zepbound dose (2.5 mg) rather than jumping to a higher dose, even if you were already at maintenance on Wegovy. This minimizes GI side effects.
Does insurance cover Zepbound or Wegovy?
Both drugs face similar insurance hurdles. Many commercial plans now cover one or both, but typically place them on Tier 3 with prior authorization requirements. Coverage criteria usually include BMI ≥ 30 (or ≥ 27 with a comorbidity) and documented prior weight loss attempts. Medicare Part D generally does not cover either drug for weight loss alone, though Wegovy can be covered for the cardiovascular indication approved in 2024. Medicaid coverage varies by state.
Which loses more weight: Wegovy or Zepbound?
Zepbound produces more weight loss on average. In SURMOUNT-1, patients on Zepbound 15 mg lost 20.9% of their body weight at 72 weeks. In STEP 1, patients on Wegovy 2.4 mg lost 14.9% at 68 weeks. The first head-to-head trial, SURMOUNT-5 (2025), confirmed the difference: tirzepatide patients lost about 6.5 percentage points more body weight than semaglutide patients over the same period. Individual results vary, and a substantial minority do equally well on either.
Is Wegovy or Zepbound easier on the stomach?
For most patients, both drugs cause similar GI side effects and both are easier to tolerate when titration is slow. Some real-world reports suggest Wegovy is marginally gentler at the start, while others find Zepbound smoother because GIP activity may modestly offset nausea. The honest answer is that there is no reliable way to predict which one your stomach will tolerate better. Many providers will pause titration or step down if side effects become severe, regardless of which drug.
Wegovy pill vs injection — is there an oral version?
Wegovy itself is only available as a once-weekly subcutaneous injection. Novo Nordisk also makes Rybelsus, an oral semaglutide tablet, but Rybelsus is FDA-approved for type 2 diabetes, not weight loss. A higher-dose oral semaglutide for obesity has completed Phase 3 trials and is under FDA review. Zepbound is also injection-only at this time, with no oral version on the market. For more on oral options, see our Wegovy pill guide.
How long do you have to be on Zepbound or Wegovy?
Both medications are intended for chronic, long-term use. Clinical trials and real-world data show that when patients stop either drug, most regain a significant share of the weight they lost within 12 months. Obesity is treated as a chronic condition, similar to high blood pressure or type 2 diabetes — the medication works while you take it and stops working when you stop. Most prescribers expect treatment to continue indefinitely as long as it remains effective and well tolerated.
Is tirzepatide a GLP-1 like semaglutide?
Tirzepatide activates the GLP-1 receptor, but it is not a pure GLP-1 agonist the way semaglutide is. Tirzepatide is a single molecule that binds to both the GLP-1 receptor and the GIP (glucose-dependent insulinotropic polypeptide) receptor. This dual action is why it is sometimes called a "twincretin". GIP is the other major incretin hormone released by the gut after eating, and adding GIP activity to GLP-1 activity appears to enhance both glucose control and weight loss in trials.